ALPROLIX® prophylaxis has extended dosing vs SHLs that helps reduce the treatment burden1,2
![]()
ALPROLIX prophylaxis offers proven protection* at dosing intervals to fit your patients’ needs
Recommended Starting Doses
7-day interval
For dosing on the same day every week1
- The recommended starting dose is 50 IU/kg for adults/adolescents (aged ≥12 years) and 60 IU/kg for children (aged ≤11 years)
- A 7-day interval is the most common EHL prophylaxis regimen in hemophilia B†
10-day interval
For less frequent dosing1
- Another recommended starting dose is 100 IU/kg for adults and adolescents
- For patients on ALPROLIX looking to extend their dosing interval beyond their initial starting interval, adjust the dose based on individual patient response
- Dose and duration of treatment depend on the severity of the factor IX deficiency, the location and extent of bleeding, the individual patient’s PK profile, and/or the patient’s clinical condition
Extended Dosing
14-day interval
Potential for ≥14-day dosing for fewer infusions2,3
- In the B-LONG and B-YOND trials, some adult and adolescent patients extended their dosing interval to ≥14 days
*ALPROLIX has been proven to help patients prevent bleeding episodes using a prophylaxis regimen.1
†Data are from an online survey conducted by Sanofi in April 2019 with 359 adult patients and caregivers to provide insights into the US hemophilia A and hemophilia B markets. Ninety respondents were adults with hemophilia B and 60 respondents were caregivers of a patient with hemophilia B.
EHL=extended half-life; PK=pharmacokinetic; SHL=standard half-life.
Clinical trial information
B-LONG was a phase 3 open-label study that investigated the safety and efficacy of ALPROLIX in 123 previously treated adult and adolescent patients aged ≥12 years with severe hemophilia B. The study included a fixed-interval (weekly) arm (n=63), a fixed-dose (interval-adjusted) arm (n=29), an episodic (on-demand) arm (n=27), and a surgical arm (n=12).1
B-YOND was an open-label extension trial that studied the long-term safety and efficacy of ALPROLIX over 5 years in 120 adult, adolescent, and pediatric patients previously treated in Kids B-LONG or B-LONG. The study included a fixed-interval arm (n=74), a fixed-dose arm (n=36), a modified prophylaxis arm (n=17), and an episodic (on-demand) arm (n=15).4
*ALPROLIX has been proven to help patients prevent bleeding episodes using a prophylaxis regimen.1
PEDIATRIC PATIENTS DOSING
With ALPROLIX, children can start on a treatment that can grow with them, as their needs may change over time1
![]()
For children aged <12 years, the recommended prophylaxis starting regimen is
60 IU/kg once weekly,
with adjustments made based on patient response1
- More frequent or higher doses may be needed in children aged <12 years, and especially in children aged <6 years
- On average, 1 IU of ALPROLIX per kg of body weight increases the level of circulating factor IX by approximately 1% in children aged ≥6 years and 0.6% in children aged <6 years
~93%
of pediatric patients maintained or extended their dosing interval through the B-YOND extension trial4
- In B-YOND (n=27), 21 patients maintained and 4 patients extended their dosing interval
ADULTS AND ADOLESCENTS DOSING
ALPROLIX offers flexibility of prophylaxis dosing intervals to fit your patients’ needs
![]()
For patients who want a regimen on the same day every week, initiate treatment at
50 IU/kg every 7 days1
For fewer infusions, ALPROLIX offers a second prophylaxis starting dose1
- ALPROLIX can be initiated at 100 IU/kg every 10 days
- To extend the dosing interval beyond a patient’s initial starting interval, adjust the dose based on individual patient response
- Dose and duration of treatment depend on the severity of the factor IX deficiency, the location and extent of bleeding, the individual patient’s pharmacokinetic profile, and/or the patient’s clinical condition
~85%
of adult and adolescent patients maintained or extended their dosing interval through the B-YOND extension trial4
- In B-YOND (n=71), 56 patients maintained and 4 patients extended their dosing interval
![]()
≥14-DAY DOSING
For patients who want extended dosing, ALPROLIX offers the potential for ≥14-day dosing
- More than half of patients (54%) in the interval-adjusted arm (n=26) extended their dosing interval to ≥14 days during the last 3 months of B-LONG2
Average dosing interval for interval-adjusted arm2
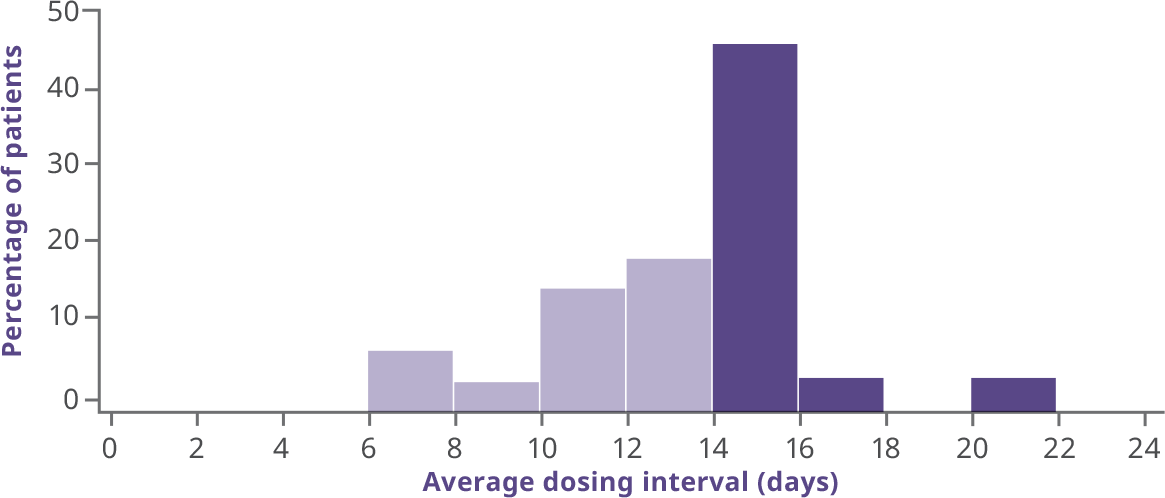
- Patients initiated treatment at 100 IU/kg every 10 days; the majority were well controlled and experienced zero bleeds before extending their interval3
- Overall median dosing interval in B-LONG was 12.5 days (10.4-13.4)1
- Additional patients were able to extend their dosing interval during the B-YOND extension trial3
Post hoc analysis: ALPROLIX prophylaxis efficacy data at ≥14-day interval (n=22)3:
-
Median overall ABR:
1.6 (0.6-2.7)
-
Median AsBR:
0.7 (0.3-1.1)
-
Median joint ABR:
1.0 (0.3-1.6)
- Data are reported from a post hoc analysis of 22 adult and adolescent patients (aged ≥12 years) who received ALPROLIX prophylaxis with a dosing interval of ≥14 days at any time during B-LONG or B-YOND, up until the time of the second interim analysis
- The analysis was not powered to show statistical significance. There was a small sample size, no control group, and potential for interobserver variability
- Patients in the Kids B-LONG study were not included in this post hoc analysis because the study design did not allow for dosing intervals longer than 1 week
In the interval-adjusted arm of B-LONG (n=29)3:
-
Median overall ABR:
1.38 (0-3.43)
-
Median AsBR:
0.88 (0-2.3)
-
Median joint ABR:
0.36 (0-3.24)
Protection,* Experience, and Dosing:
A Discussion With
Dr De Angulo
With the widest range of vial strengths in hemophilia
B,
ALPROLIX offers the potential to resolve bleeds with
fewer
vials1,5-10

ABR=annualized bleed rate; AsBR=annualized spontaneous bleed rate.
 WATCH NOW:
WATCH NOW: 

